A00-281 Practice Exam - SAS Clinical Trials Programming Using SAS 9 - Accelerated Version 9 Accelerated Version
Reliable Study Materials & Testing Engine for A00-281 Exam Success!
Exam Code: A00-281
Exam Name: SAS Clinical Trials Programming Using SAS 9 - Accelerated Version 9 Accelerated Version
Certification Provider: SAS Institute
Corresponding Certifications: Clinical Trials Programmer , SAS Certified Clinical Trials Programmer Using SAS 9

Free Updates PDF & Test Engine
Verified By IT Certified Experts
Guaranteed To Have Actual Exam Questions
Up-To-Date Exam Study Material
99.5% High Success Pass Rate
100% Accurate Answers
100% Money Back Guarantee
Instant Downloads
Free Fast Exam Updates
Exam Questions And Answers PDF
Best Value Available in Market
Try Demo Before You Buy
Secure Shopping Experience
A00-281: SAS Clinical Trials Programming Using SAS 9 - Accelerated Version 9 Accelerated Version Study Material and Test Engine
Last Update Check: Mar 19, 2026
Latest 99 Questions & Answers
45-75% OFF
Hurry up! offer ends in 00 Days 00h 00m 00s
*Download the Test Player for FREE
Dumpsarena SAS Institute SAS Clinical Trials Programming Using SAS 9 - Accelerated Version 9 Accelerated Version (A00-281) Free Practice Exam Simulator Test Engine Exam preparation with its cutting-edge combination of authentic test simulation, dynamic adaptability, and intuitive design. Recognized as the industry-leading practice platform, it empowers candidates to master their certification journey through these standout features.
What is in the Premium File?
Satisfaction Policy – Dumpsarena.co
At DumpsArena.co, your success is our top priority. Our dedicated technical team works tirelessly day and night to deliver high-quality, up-to-date Practice Exam and study resources. We carefully craft our content to ensure it’s accurate, relevant, and aligned with the latest exam guidelines. Your satisfaction matters to us, and we are always working to provide you with the best possible learning experience. If you’re ever unsatisfied with our material, don’t hesitate to reach out—we’re here to support you. With DumpsArena.co, you can study with confidence, backed by a team you can trust.
SAS Institute A00-281 Exam FAQs
Introduction of SAS Institute A00-281 Exam!
The SAS Certified Clinical Trials Programmer Using SAS 9 exam (A00-281) is a certification exam offered by SAS Institute. It is designed to assess the knowledge and skills of professionals who develop and maintain clinical trials programs using SAS software. The exam covers topics such as SAS programming, data management, and clinical trial design.
What is the Duration of SAS Institute A00-281 Exam?
The SAS Institute A00-281 exam is a two-hour exam.
What are the Number of Questions Asked in SAS Institute A00-281 Exam?
There are 60 questions in the SAS Institute A00-281 exam.
What is the Passing Score for SAS Institute A00-281 Exam?
The passing score for the SAS Institute A00-281 exam is 70%.
What is the Competency Level required for SAS Institute A00-281 Exam?
The SAS Institute A00-281 exam is an advanced-level certification exam. It is designed to assess the knowledge and skills of experienced SAS professionals who have a minimum of two years of experience working with SAS software. To pass the exam, candidates must demonstrate a comprehensive understanding of SAS programming, data manipulation, and analytics.
What is the Question Format of SAS Institute A00-281 Exam?
The SAS Institute A00-281 exam consists of multiple choice and fill-in-the-blank questions.
How Can You Take SAS Institute A00-281 Exam?
The SAS Institute A00-281 exam can be taken online or in a testing center. To take the exam online, you will need to create an account on the SAS Institute website and register for the exam. Once you have registered, you will be given instructions on how to access the exam. To take the exam in a testing center, you will need to contact the testing center to find out what their procedures are for taking the exam.
What Language SAS Institute A00-281 Exam is Offered?
The SAS Institute A00-281 exam is offered in English.
What is the Cost of SAS Institute A00-281 Exam?
The cost of the SAS Institute A00-281 exam is $180 USD.
What is the Target Audience of SAS Institute A00-281 Exam?
The target audience for the SAS Institute A00-281 exam is individuals who want to become SAS Certified Data Scientists. This certification exam is intended for professionals with experience in data science, analytics, and predictive modeling. Professionals looking to validate their knowledge and skills in data science, analytics, and predictive modeling should consider taking this exam.
What is the Average Salary of SAS Institute A00-281 Certified in the Market?
The average salary in the market after obtaining SAS Institute A00-281 certification varies widely depending on the country and the job title. Generally, the average salary range for certified SAS professionals is between $60,000 and $90,000 per year.
Who are the Testing Providers of SAS Institute A00-281 Exam?
SAS Institute does not provide an official testing provider for the A00-281 exam. However, there are various third-party providers that offer practice tests and study materials to help individuals prepare for this exam. Popular providers include SAS Institute, PrepAway, and Exam-Labs.
What is the Recommended Experience for SAS Institute A00-281 Exam?
The recommended experience for the SAS Institute A00-281 exam includes familiarity with SAS Enterprise Guide, SAS Enterprise Miner, SAS Visual Analytics and SAS Grid Manager. Additionally, hands-on experience in developing, validating and deploying analytical models, and knowledge in the areas of data preparation, data exploration, descriptive statistics and predictive analytics is also recommended.
What are the Prerequisites of SAS Institute A00-281 Exam?
The prerequisite for the SAS Institute A00-281 exam is a fundamental knowledge of SAS programming and experience with the SAS software. It is recommended that candidates have at least six months of experience with the SAS software before attempting the exam.
What is the Expected Retirement Date of SAS Institute A00-281 Exam?
The official website for SAS Institute A00-281 exam is https://www.sas.com/en_us/certification/exam-details/a00-281.html. It does not provide information on the expected retirement date of the exam.
What is the Difficulty Level of SAS Institute A00-281 Exam?
The difficulty level of the SAS Institute A00-281 exam is moderate.
What is the Roadmap / Track of SAS Institute A00-281 Exam?
The SAS Institute A00-281 exam is a certification track/roadmap for SAS Certified Clinical Trials Programmer Using SAS 9. This exam is designed to test a candidate’s knowledge and skills related to the design, implementation, and analysis of clinical trials using SAS 9. The exam covers topics such as SAS programming, data management, statistical analysis, and reporting. Candidates who pass the exam will earn the SAS Certified Clinical Trials Programmer Using SAS 9 credential.
What are the Topics SAS Institute A00-281 Exam Covers?
The SAS Institute A00-281 exam covers the following topics:
1. SAS Programming Language: This section covers topics related to the SAS programming language, such as data manipulation, data analysis, and data management.
2. SAS Data Management: This section covers topics related to the management of SAS data, such as data access, data manipulation, and data analysis.
3. SAS Visual Analytics: This section covers topics related to the use of SAS Visual Analytics, such as data exploration, data visualization, and data analysis.
4. SAS Business Intelligence: This section covers topics related to the use of SAS Business Intelligence, such as data analysis, data visualization, and data governance.
5. SAS Enterprise Miner: This section covers topics related to the use of SAS Enterprise Miner, such as data mining, data analysis, and model building.
6. SAS Certification: This section covers topics related to the SAS certification process, such as the exam
What are the Sample Questions of SAS Institute A00-281 Exam?
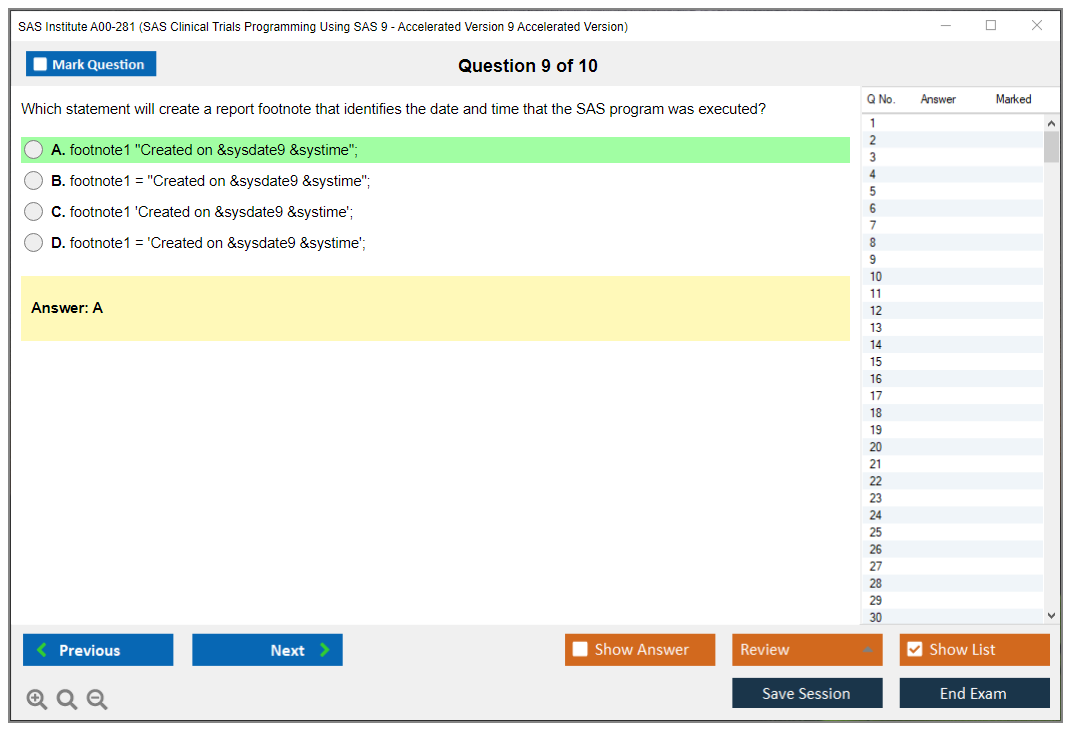
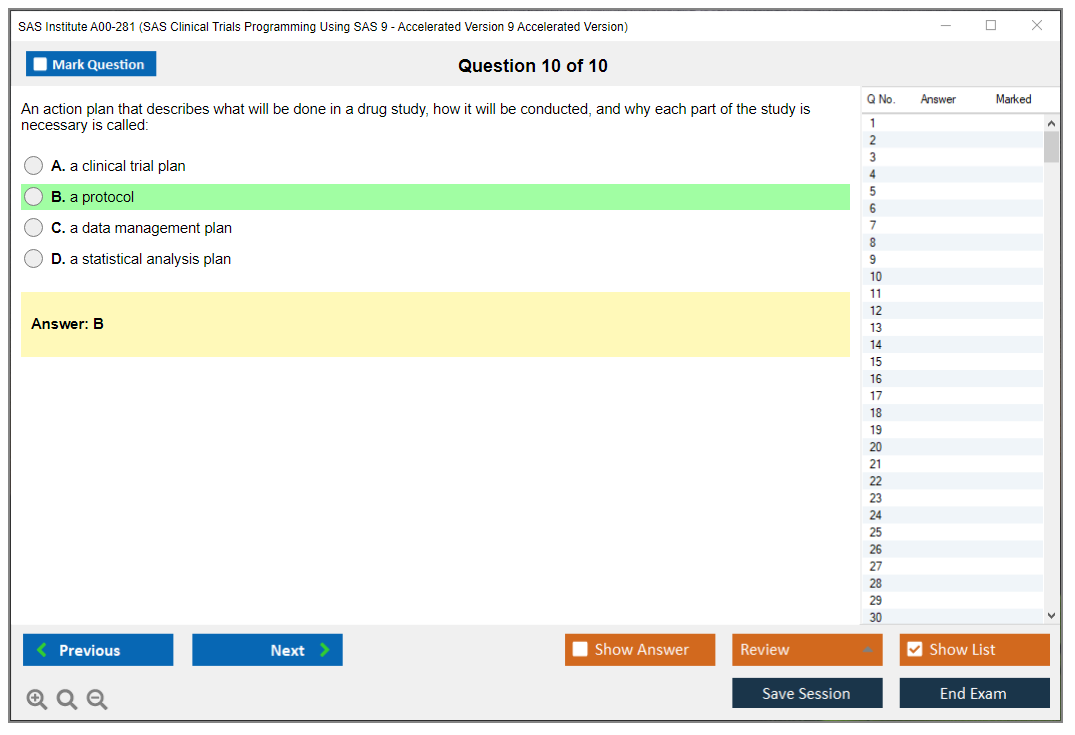
1. What is the purpose of the SAS/ACCESS interface?
2. What is the syntax used to define a macro variable?
3. How can you create a data set using a SAS DATA step?
4. What is the purpose of the SAS SQL procedure?
5. How can you create a report using the SAS REPORT procedure?
6. What is the difference between SAS functions and SAS procedures?
7. What is the purpose of the SAS Autocall facility?
8. What are the different types of SAS indexes?
9. How can you create a graph using the SAS Graph procedure?
10. What is the purpose of the SAS Macro Language?
Understanding the SAS Institute A00-281 Exam: SAS Clinical Trials Programming Using SAS 9 - Accelerated Version What makes A00-281 different from your typical SAS certification The SAS A00-281 exam isn't just another programming test. This is the accelerated version of clinical trials programming, and it's designed for people who already know their way around SAS but need to break into the pharma world fast. The standard A00-280 certification takes you through clinical programming step by step, but the A00-281 version assumes you've got solid programming chops and just need to learn the clinical piece. Makes the whole thing way more intense. The pharmaceutical industry runs on clinical trials data. Every drug you see at the pharmacy went through years of trials. Someone had to program those datasets, create analysis files, and generate reports that regulatory agencies like the FDA could actually understand. That's where SAS Clinical Trials Programming Using SAS 9 comes in. The A00-281... Read More
Understanding the SAS Institute A00-281 Exam: SAS Clinical Trials Programming Using SAS 9 - Accelerated Version
What makes A00-281 different from your typical SAS certification
The SAS A00-281 exam isn't just another programming test. This is the accelerated version of clinical trials programming, and it's designed for people who already know their way around SAS but need to break into the pharma world fast. The standard A00-280 certification takes you through clinical programming step by step, but the A00-281 version assumes you've got solid programming chops and just need to learn the clinical piece. Makes the whole thing way more intense.
The pharmaceutical industry runs on clinical trials data. Every drug you see at the pharmacy went through years of trials. Someone had to program those datasets, create analysis files, and generate reports that regulatory agencies like the FDA could actually understand. That's where SAS Clinical Trials Programming Using SAS 9 comes in. The A00-281 certification proves you can handle this work, not just basic data manipulation.
What's strange is how specific this gets. You're not just joining tables and running PROC MEANS. You're creating SDTM domains from raw case report forms, building ADaM analysis datasets that statisticians actually want to use, and programming tables that meet regulatory submission standards. The accelerated format crams what's normally spread across multiple courses into one intensive certification path. Short answer, long preparation.
Who actually needs this credential and why
Clinical SAS programmers are the obvious candidates. If you're working at a pharma company or CRO and touching clinical data, this certification makes you way more valuable. I've seen job postings that specifically mention A00-281 or similar clinical programming credentials because hiring managers want proof you understand the domain, not just that you can write code.
Biostatisticians sometimes take this route too. Stats people often know the analysis side cold but struggle with the data prep and submission programming. Getting the SAS clinical trials programming exam under your belt bridges that gap. Same deal for data managers who want to move beyond database design into actual programming roles. It's the natural next step for a lot of them.
QC programmers need this knowledge desperately. You're validating someone else's work, which means you need to understand clinical programming standards inside and out. The A00-281 certification validates you know what correct clinical programming actually looks like.
Regulatory affairs folks?
They occasionally pursue this too, though they're not usually doing hands-on programming. Understanding how clinical datasets get built and what goes into those massive regulatory submissions helps them do their job better. Plus it gives them credibility when they're talking to programming teams. I once worked with a regulatory specialist who learned just enough SAS to be dangerous at interdepartmental meetings. She couldn't write production code but she could spot when programmers were bullshitting about timelines, which honestly made everyone's life easier in the long run.
The real-world skills this exam actually tests
Creating and manipulating clinical trial datasets sounds straightforward until you're actually doing it. The SAS 9 clinical programming certification tests whether you can take messy raw data from a clinical trial and turn it into standardized CDISC SDTM SAS programming structures. Every domain has specific requirements. Adverse events look different from lab results, which look different from efficacy endpoints. You've gotta memorize a lot of these distinctions.
Then there's ADaM dataset creation in SAS. This is where things get interesting. Analysis Data Model datasets are what statisticians use for their actual analyses. You're deriving variables, creating flags, handling missing data, and making sure everything traces back to the SDTM source. One mistake here and the entire statistical analysis plan falls apart.
Programming clinical listings tables figures (TLFs) SAS is probably what you'll spend the most time doing in actual jobs. PROC REPORT becomes your best friend. You need to format things exactly right because these tables end up in clinical study reports that go to regulatory agencies. The spacing matters. The footnotes matter. Even the font choices can matter depending on submission requirements.
SAS clinical data validation and QC workflows are huge on this exam. Double programming is standard in pharma. Two programmers independently create the same output and then compare results. You need to know how to set up these validation processes and document everything properly, which feels tedious but it's how you catch mistakes before they reach regulators.
How the accelerated format changes your prep strategy
The accelerated version assumes you're already comfortable with SAS Base Programming concepts. DATA steps? PROC SQL? Basic macros? All that should be second nature. If you're still looking up basic syntax, you should probably start with A00-215 or A00-211 first and build that foundation. Otherwise you'll struggle.
What the acceleration really means is they integrate basic and advanced clinical concepts faster. Standard certifications might spend weeks on SDTM before touching ADaM. The A00-281 exam expects you to understand both and how they relate to each other pretty much immediately. The study materials are denser. The practice scenarios are more complex. There's less hand-holding overall.
For experienced programmers coming from other industries, this is actually perfect. You don't need someone explaining what a merge is for the hundredth time. You need to understand what makes clinical data special and how to apply your existing skills in this new domain.
Understanding exam logistics and what to expect
The SAS A00-281 exam cost typically runs around $180, though pricing can vary by region and whether your employer has any corporate agreements with SAS Institute. You take it at Pearson VUE testing centers, which are everywhere. There's probably one within 30 minutes of wherever you're reading this.
It's a closed-book, computer-based exam. You're not getting access to SAS documentation during the test, which means you need to actually memorize syntax and conventions. The questions mix multiple-choice with scenario-based problems where they describe a programming situation and ask you to identify the correct approach.
Passing score?
SAS Institute doesn't always publicly detail specifics, but generally you're looking at needing roughly 68-70% correct to pass. They use scaled scoring, so the exact number of questions you need to get right can vary slightly based on question difficulty. Not rocket science but not a gimme either.
How A00-281 fits with other clinical programming credentials
If you've already got A00-212 advanced programming certification, the transition to A00-281 makes total sense. You've got the technical foundation, now you're adding the clinical piece. Some people go the opposite direction. Get the clinical cert first, then go back for advanced programming to round out their skills.
The relationship with CDISC certifications? It's interesting. SAS Institute focuses on the programming implementation, while CDISC certifications cover the standards themselves more broadly. Having both makes you extremely marketable because you understand not just how to program the standards but why they exist and how they evolved.
For career progression, A00-281 opens doors to senior clinical programmer roles, lead programmer positions, and eventually programming management. It's also a stepping stone if you want to move into statistical programming, clinical data science, or specialized areas like medical writing support.
Some people combine this with A00-251 for administration skills or A00-260 for data integration, creating a broader skill set that's valuable in larger pharma organizations where you might touch multiple aspects of the data pipeline.
The CDISC standards piece you can't ignore
CDISC SDTM SAS programming is absolutely central to this exam. Study Data Tabulation Model isn't just a nice-to-have. It's the regulatory standard for submitting clinical trial data. You need to understand the different domain classes, required and expected variables, and controlled terminology.
The exam tests whether you can look at raw clinical data and know which SDTM domain it belongs in. Adverse events go in AE, demographics in DM, exposure in EX. But then you've got supplemental qualifiers, timing variables, and all these details that matter when regulators are reviewing your submission.
ADaM is the analysis side. You're creating datasets that follow specific structures like ADSL (subject-level), ADAE (adverse events analysis), and BDS (basic data structure) for efficacy endpoints. These aren't arbitrary structures. They're designed to make statistical analysis reproducible and transparent, which sounds straightforward but gets complicated fast when you're dealing with complex trial designs.
Study materials that actually help versus marketing fluff
The official SAS training courses for A00-281 are solid but expensive. We're talking thousands of dollars for the full accelerated track. If your employer pays, great. If not, you need to be strategic about what you actually need versus what's nice to have.
SAS documentation? Free and thorough. The SAS Institute A00-281 study guide materials often reference specific procedures and techniques you can look up in detail. Spend time with PROC REPORT documentation especially. That procedure does heavy lifting in clinical programming.
Third-party resources vary wildly in quality. Some A00-281 practice test materials are outdated or focus on wrong topics. Look for resources that explicitly mention SAS 9.4 and recent CDISC standards. Anything talking about SAS 9.1 or 9.2 is probably too old to be useful.
Hands-on practice matters more than reading. Set up practice scenarios where you create SDTM domains from scratch, build ADaM datasets, and generate TLFs. The muscle memory of actually writing the code helps way more than just reading about it. Sounds obvious but people still try to pass this by memorizing slides.
Renewal and staying current in clinical programming
Here's the thing: SAS certifications don't technically expire, but the field moves fast enough that a certification from 2015 doesn't carry the same weight as a recent one. Clinical data standards change. New procedures get added to SAS. Regulatory expectations shift.
Most serious clinical programmers recertify or pursue additional credentials every few years. You might add A00-277 for visual analytics skills or branch into A00-225 for predictive modeling if you're moving toward more advanced analytics roles.
The transition from SAS 9 to Viya? It's happening across the industry, though SAS 9 isn't going anywhere soon in pharma. Clinical trials that started years ago still need SAS 9 programmers to finish them out. But learning Viya programming through certifications like A00-415 positions you well for the future.
Staying current also means following CDISC updates, FDA guidance documents, and industry best practices. The certification gets you in the door, but continuous learning keeps you relevant in an industry where regulatory requirements and data standards constantly shift. Keeps the work interesting even after you've been doing it for years, which beats the alternative of stagnating in a role that automation will eventually replace anyway.
A00-281 Exam Cost, Registration, and Logistics
What this certification proves
The SAS A00-281 exam is basically SAS saying, "yes, this person can survive real clinical programming work in SAS 9." It maps to SAS Clinical Trials Programming Using SAS 9 on the accelerated track, so the vibe is practical and job-facing. Not academic theory.
You're expected to be comfortable with clinical trial data programming tasks that show up in sponsor or CRO pipelines. Think CDISC SDTM SAS programming, basic ADaM dataset creation in SAS, and producing clinical listings tables figures (TLFs) SAS without melting down when the spec changes mid-sprint.
Who should take it (and who should wait)
This exam fits junior to mid clinical programmers, SAS programmers moving into pharma, and data folks who already do SAS but need the credential for hiring filters.
Absolute beginners? Wait.
Honestly, if you haven't written a DATA step from scratch, read a log for warnings, and built something like a listing with proc report proc sql clinical programming, you're gonna spend most of the test translating the question instead of answering it. That's a bad use of exam money.
What you'll pay in 2026
People ask, "How much does the SAS A00-281 exam cost?" The realistic answer for 2026 is: plan for the usual SAS range, then verify the current price right before checkout. Standard SAS Institute exam pricing is typically $180 to $250 USD, subject to updates, and yes, they do change it sometimes without much drama.
Regional pricing variations are real. If you register from certain countries, you may see local currency pricing that isn't a straight conversion. Taxes can get added depending on where you live, so your final number might land higher than you expected even if the base exam fee looks normal on paper. This happens especially in places with VAT or similar regulations that get tacked on at the very end of checkout.
Discounts exist. They're not magic.
If you're part of the SAS Global Certification Program membership or you have some official discount path through SAS, you might see reduced pricing. Worth checking before you pay full price because even a small discount is still money back in your pocket.
Academic pricing can also apply if you're enrolled in an accredited institution. Look, don't assume you qualify just because you have a .edu email. You usually need to follow their academic verification rules, and sometimes the discount applies through a specific academic store path rather than the standard certification checkout. I mean, it's annoying, but it's how they control eligibility.
Corporate volume pricing is a thing too. If your org is certifying multiple employees, ask your training coordinator or procurement team to talk to SAS about bulk options. The per-exam cost can come down, and you might also get centralized voucher handling which makes the whole process less annoying.
Retakes cost money. Usually you pay again per attempt unless you're on some promo, and your retake fee is typically the same as the exam price or close enough that you should budget as if it's full price.
Signing up without getting lost
The registration process for the A00-281 certification starts with creating a SAS profile. You'll use that to access the SAS certification portal, pick the exam, and then jump out to the delivery vendor for scheduling.
Next step: select A00-281 certification from the clinical programming options. The naming can be a little clunky, so double-check the full title to avoid registering for the wrong test. SAS has multiple clinical and programming exams that sound similar if you skim.
Then you choose delivery: Pearson VUE testing center or online proctoring. Both work, but the choice changes your exam day stress in totally different ways. I'll get into that below.
Scheduling flexibility depends on your region and whether you're doing online. Testing centers have limited seats. Online has more time slots, but proctors can be strict and tech issues are a real risk, so "more slots" doesn't always mean "more convenient."
You'll need required ID documents. Usually that means a valid government-issued photo ID and matching name details. Name mismatches are a dumb way to lose an exam appointment, so fix your profile before you schedule.
After you register, you'll get confirmation emails with the appointment details and policy links. Sometimes you also get pointers to prep materials, but don't expect a full SAS Institute A00-281 study guide to land in your inbox automatically. You still need to go gather resources.
Booking rules you should treat seriously
Book earlier than you think. The sane recommendation is 2 to 4 weeks ahead of your desired date, especially if you need a specific day of week, or you're scheduling around work travel, or you want a particular testing center.
Peak testing periods happen. End of quarter. End of year. Also right before big hiring waves or internship timelines.
Seats vanish.
Rescheduling usually has a deadline, commonly 24 to 48 hours before the exam. Miss it and you can lose the fee. Cancellation policies can be similar, with refunds depending on timing and local rules, so read the policy at checkout instead of assuming it's like airline tickets.
Emergency rescheduling exists, but it's not a guaranteed kindness. If you're sick, have a family emergency, or your testing center shuts down, you'll typically need documentation and you'll be dealing with support queues. Keep screenshots and emails.
Time zone issues hit online proctored exams all the time. Your appointment time might display in one zone during booking and another in your confirmation. Verify it the day before, not ten minutes before.
Testing center vs online proctoring
Pearson VUE testing centers are the classic setup: controlled room, consistent hardware, and fewer weird interruptions. You show up, lock your stuff away, and you take the exam. It's boring, which is good. The biggest advantage is minimal technical drama, and honestly that alone is worth it if your home internet is flaky.
Online proctoring is convenient, but picky. You need a webcam, microphone, stable internet, and a private space. No second monitor. No random people walking in. No "my roommate is cooking behind me." The proctoring software may require installation and permissions that your work laptop might block. Privacy concerns are fair because you're literally being recorded while someone watches you through the camera for the entire duration of your test session.
Do the system check early. Like days early. Run the compatibility test, verify camera and mic, and test the exact room lighting. If something fails on exam day, support might help, but you can still lose your slot if you can't get compliant fast enough.
Once I tried testing online while a construction crew started jackhammering outside my window at 9 AM. The proctor kept asking if I was being disturbed. Yeah, obviously. But pausing meant losing time, so I just powered through while my desk vibrated. Not ideal. Testing centers don't usually come with surprise roadwork.
Retakes and how to think about them
Waiting periods commonly apply, often around 14 days between attempts. Also, there can be limits on the number of retakes within a set timeframe, so don't treat it like unlimited respawns.
Cost adds up fast. Two attempts at $200-ish is suddenly "I could have paid for a real prep course" money. It's smarter to plan your first attempt like it's the only one.
Score reports usually show domain-level performance, not every question you missed. That's still useful. If you bombed clinical standards concepts, go back and drill SDTM and ADaM basics, plus SAS clinical data validation and QC workflows. A lot of exam misses come from sloppy assumptions, not syntax.
When should you train vs retake quickly? If you were close and you know you panicked, a quick retake makes sense. If you were lost on half the blueprint, pause and do targeted training and an A00-281 practice test style review. Otherwise you're paying to feel bad twice.
Exam day: what to bring and what not to do
Bring valid government-issued photo identification. The name must match your registration.
No excuses.
Leave prohibited items behind. Notes, phones, smartwatches, papers, and study materials are usually banned. Some centers are strict about jewelry and pockets too, so wear simple stuff.
Arrive 15 to 30 minutes early for a testing center. You'll do check-in, security steps, maybe a photo, maybe a palm vein scan depending on location, then you'll be seated.
They provide scratch paper or a whiteboard style note sheet and a pen or marker. Calculator rules vary by exam, so don't assume you can bring one.
Break policies depend on exam length. If breaks are allowed, they're often timed or the clock keeps running. Plan your pacing like you might not get a true pause.
Accommodations and special situations
If you need accessibility accommodations, request them before scheduling if possible. You'll usually need documentation, and approval can take time. Don't leave it to the last week.
Extended time is common for approved cases. Alternative formats may exist if the standard delivery doesn't work for you.
Language assistance is limited. Most candidates take the exam in English. If you're a non-native speaker, your best move is extra practice reading question wording fast. Clinical questions love long setups.
Religious or cultural accommodation requests can be possible, but you need to ask early and get it documented in the booking notes. Not mentioned casually on exam day.
International candidates: the extra logistics
Exam availability varies by country. Some regions have fewer testing centers, so you might end up traveling. That adds cost and scheduling complexity.
Payment methods can vary too. Currency conversion is not always friendly, and some cards get blocked on international transactions. Have a backup card or payment option ready.
SAS certifications are generally recognized across borders, but how they're valued depends on local hiring norms and regulatory environments. In some markets, the credential is a checkbox. In others, your portfolio of SAS clinical trial data programming work and your ability to explain QC decisions matters more.
Quick FAQs people keep Googling
What is the passing score for A00-281? SAS doesn't always publish a simple universal number, and passing rules can be versioned. Check the current exam page for scoring details.
Is SAS A00-281 difficult for beginners? Yes. Not impossible, but it's a rough first SAS exam if you haven't done clinical-style datasets and TLF outputs.
What are the best study materials for SAS Clinical Trials Programming Using SAS 9? SAS training for the accelerated track, SAS docs for the procedures you actually use, and hands-on reps building SDTM-ish structures and listings.
Are there official A00-281 practice tests and how accurate are they? Sometimes there are official prep tools, sometimes not. If you use third-party sets, treat them as reading and timing practice. Not as an answer key to the real exam.
The thing is, if you want, I can also write a separate post that's purely about how to pass SAS A00-281, with a week-by-week plan and what to drill for SDTM, ADaM, and TLFs.
What is the Passing Score for A00-281 and How Scoring Works
Look, if you're gearing up for the SAS A00-281 exam, you need to know what score actually gets you across the finish line. The passing score for the SAS Clinical Trials Programming Using SAS 9 - Accelerated Version typically hovers around 68-70% correct responses, though SAS Institute doesn't publish exact cutoffs publicly and these thresholds can shift slightly depending on exam version. Not gonna lie, this ambiguity drives people crazy. But there's actually solid reasoning behind how SAS handles scoring.
Why SAS uses scaled scoring instead of raw percentages
Here's the thing about standardized exams. SAS doesn't just count up your right answers and slap a percentage on it. They use scaled scoring methodology, which means your raw score (the actual number you got correct) gets converted through statistical analysis to a scaled score. I mean, think about it: if one exam version happens to be slightly harder than another, it wouldn't be fair holding everyone to the exact same raw score requirement, would it?
The equating process makes sure that someone taking version A faces the same difficulty as someone taking version B months later. SAS employs psychometric methods analyzing question performance across thousands of test-takers, adjusting the passing threshold so difficulty variations don't penalize candidates unfairly. Your final score might appear on a scale from 200-800 (common for many IT certifications) or as a percentage, but either way, that number reflects performance normalized across all exam versions.
Reminds me of how my old university curved organic chemistry exams after everyone bombed the thermodynamics section. Same principle, just more sophisticated statistics.
What you're actually facing question-wise
The A00-281 exam throws approximately 60-70 questions at you. All multiple-choice. Single best answer. No partial credit here, you either nail it or you don't. Every question carries equal weight unless SAS explicitly states otherwise in the exam blueprint, which they rarely do for this certification.
You'll encounter scenario-based questions presenting realistic clinical programming situations, stuff like "Given this SDTM dataset structure, which approach correctly derives the baseline flag?" Code interpretation questions test whether you can read SAS syntax and predict what it'll actually do when executed. Output analysis questions might show you PROC REPORT results and ask you to identify errors or explain derivation logic. The exam pulls from clinical data standards, data manipulation techniques, TLFs programming, and validation practices based on the published blueprint.
Honestly, the mix keeps you on your toes.
Some questions take 30 seconds. Others require careful reading of scenario details and mentally tracing through DATA step logic, which can eat 3-4 minutes easily.
Time management when you've got 105-120 minutes
You're looking at 105-120 minutes total depending on your exam version. That breaks down to roughly 1.5-2 minutes per question on average. Sounds generous until you hit a complex scenario with multiple dataset merges and conditional logic to break down, right? The computer-based testing system lets you mark questions for review, which is clutch when you're stuck.
There's no penalty for guessing, so answer everything. I mean every single question. Leave nothing blank whatsoever. If you're running out of time, educated guesses beat empty responses 100% of the time. The system includes time tracking so you can monitor your pace, but here's my take: don't obsess over the clock every 30 seconds. Check it maybe every 15-20 questions to make sure you're not falling behind schedule.
Getting your results and what they actually tell you
You get preliminary results immediately after the computer-based exam ends.
Official score reports hit your email within 24-48 hours with way more detail. The report shows your overall scaled score, pass/fail status, and a breakdown by exam domain showing where you performed well versus where you struggled.
For the A00-281 Practice Exam Questions Pack, you're basically getting a preview of this reporting structure since quality practice materials mirror the real exam's domain distribution. The domain-level breakdown is incredibly useful. You might see that you crushed data manipulation questions but bombed on validation and QC practices. This diagnostic information becomes your roadmap if you need to retake.
You won't get question-by-question results though. SAS protects exam security by not revealing which specific questions you missed. The domain scores give you directional guidance like "scored 45% in TLFs programming" without exposing actual test content.
What domain scores reveal about your preparation
Each domain on the A00-281 has specific performance metrics in your score report. Clinical data standards and structures. Data manipulation and derivations. TLFs programming. Validation and QC practices. They're all scored separately. You need minimum competency across all domains, not just an overall passing average.
This is where people trip up sometimes. You could ace the programming questions but if you're weak on CDISC concepts or validation workflows, that'll drag down your domain scores and potentially tank your overall result. Use these metrics strategically. If you see low scores in TLFs programming, you know exactly where to focus for a retake, maybe more practice with PROC REPORT, PROC SQL for complex outputs, or understanding clinical reporting conventions better.
The A00-280 exam (the non-accelerated version) covers similar domains but at a different pace, so if you're struggling with the accelerated format, understanding the relationship between these certifications helps. Same core knowledge. Different intensity.
When things don't go as planned
Failed attempts happen. Period. You get detailed diagnostic feedback showing which domains tanked your score. SAS has retake eligibility rules and waiting periods, usually 14 days between attempts, though policies can change so verify current requirements.
Build a targeted study plan based on weak domains. If validation and QC scored low, spend time on error checking methodologies, compare procedures, and documentation standards in clinical programming contexts. Don't just re-read the same materials though. Find different resources that explain concepts from new angles.
Second attempts with focused preparation show much higher success rates. The psychological piece matters too. Don't spiral after a failed attempt. Most successful clinical programmers failed at least one certification exam. It's feedback, not failure.
For thorough preparation, the A00-281 Practice Exam Questions Pack at $36.99 gives you realistic question exposure across all domains. Combined with hands-on programming practice and solid understanding of clinical data standards, you're positioning yourself for success.
Building on foundational certifications
If A00-281 feels overwhelming, consider whether you've got the prerequisites locked down. The A00-211 (SAS Base Programming) and A00-212 (SAS Advanced Programming) establish core DATA step, PROC SQL, and macro programming skills that A00-281 assumes you already have mastered. You can technically attempt A00-281 without these certifications, but honestly, your life will be easier with that foundation solid.
The accelerated version compresses content. It's designed for experienced SAS programmers moving into clinical trials work, not beginners learning SAS and clinical concepts at the same time. Know where you stand skill-wise before diving in. The thing is, overconfidence costs you time and retake fees.
Your certificate arrives typically 2-4 weeks after passing, and you'll receive a credential number that validates your certification. The A00-281 Practice Exam Questions Pack helps you avoid the retake cycle by identifying knowledge gaps before you sit for the real thing, making that $36.99 investment look pretty smart compared to exam retake fees.
Is SAS A00-281 Difficult? Exam Difficulty Assessment
SAS A00-281 is one of those exams that sounds "just programming" until you actually look at what they expect you to know about clinical trials. Then it clicks. This is an intermediate-to-advanced test for SAS programmers, and the accelerated version especially assumes you already move fast and don't need hand-holding.
What this cert proves (and what it doesn't)
The SAS A00-281 exam is basically SAS Institute saying you can function in a clinical programming workflow, not just write clean DATA steps in a vacuum. You're being tested on your ability to read a spec, understand the clinical context, create datasets aligned to standards, and output production-style tables, listings, and figures.
Not a beginner badge. More like "job-ready" signal.
And honestly, the hardest part? It blends two skill sets that don't naturally grow together unless you've been on a study. Pure SAS people often underestimate the clinical side. Clinical folks sometimes overestimate how far "I can copy a macro" gets you. I've seen programmers with years of retail analytics experience get tripped up by a simple adverse event table because they didn't know what "treatment-emergent" meant in practice.
Who should sit for it
If you've done 6 to 12 months of SAS clinical trial data programming, you're in the sweet spot. Coming from general analytics, finance, or ops reporting? You can still pass, but you'll feel the CDISC and regulatory stuff fighting you the whole time.
Brand new to clinical programming? Look, it's doable, but it's a significant challenge. Clinical terminology, submission expectations, and the whole "this variable has a controlled terminology and a reason it exists" mindset: that's a learning curve beginners don't expect.
Pricing and scheduling details you should know
Cost-wise, SAS exam pricing can vary by region and delivery method, but most candidates see something in the ballpark of a few hundred USD. Sometimes your employer has vouchers. Sometimes SAS runs promos. Don't assume the price you saw in an old forum post is still current.
Registration is through SAS's exam provider pipeline (SAS site pushes you to the scheduling vendor). Retake policies can change, so check the current terms before you plan a "first attempt as a practice run" strategy, because that can get expensive fast.
Scoring and question format (what you can plan around)
SAS doesn't always publish every scoring detail in a way that makes test-takers happy. Passing scores can be expressed as scaled scores, and the exact cut line isn't something you can reverse-engineer.
Expect roughly 60 to 70 questions. Time pressure's real.
You'll see scenario-based items where you read a mini-spec or a data situation, then decide what code or approach matches expectations. You'll also get code review style questions where one tiny detail matters: like a BY-group assumption, a merge behavior, a retained value, or a partial date rule.
Difficulty level and pass-rate reality
Overall difficulty: intermediate to advanced for SAS programmers.
More challenging than Base SAS. Still very passable.
A lot of people ask about pass rates. You'll commonly hear ranges like 60 to 75% for adequately prepared candidates, and that lines up with what I see anecdotally in clinical programming circles: the people who actually practice with SDTM-ish structures and build TLFs under time constraints do fine, and the people who "read a guide" tend to get surprised.
The thing is, the accelerated version assumes a faster learning curve and a stronger foundation. That means less gradual progression. Topics can jump from basic domain structure into more advanced derivations without giving you a long runway, and not gonna lie, that whiplash is where good programmers still lose points.
Why beginners struggle (especially without clinical exposure)
Is the SAS clinical trials programming exam difficult for beginners? Yes, usually.
The programming isn't always the blocker. The context is.
If you've never dealt with adverse events, concomitant meds, efficacy endpoints, visit windows, or the idea of "analysis flags" being audited, you'll spend mental energy just decoding the question. Add CDISC standards on top and suddenly you're learning a new language: SDTM domain expectations, ADaM conventions, controlled terminology, and why a variable exists even if it looks redundant.
Self-study can work, but structured training helps a lot for newcomers because you need someone to connect the dots between "what the code does" and "why the study needs it that way."
The SAS programming parts that actually bite people
This isn't just PROC PRINT with a title. The SAS 9 clinical programming certification angle means you need to be comfortable building outputs and datasets that look like real deliverables.
Common technical pain points:
- Advanced DATA step work with complex conditional logic, retained values, BY-group processing, and careful merges (this is where small mistakes explode).
- proc report proc sql clinical programming for TLFs, including grouping behavior, ordering, missing handling, and producing exactly what a spec's asking for, not what you wish it asked for.
- Macro programming for reusable utilities. Not every question's heavy macro logic, but you need to read macro code and predict behavior.
- Arrays for lab/vitals reshaping and transformations, especially when the structure shifts between "wide-ish collection" and "long-ish analysis."
- Hash objects and advanced structures can show up occasionally. If you've never used them, at least understand the use case and basic mechanics.
- Debugging. SAS logs. Warnings versus errors. The exam loves testing whether you can spot what breaks in production.
If I had to explain just one in detail, it's TLF formatting. Clinical listings tables figures (TLFs) SAS questions often hide the trick in spacing, pagination logic, BY-group breaks, or ordering rules, and you can't brute-force that with "I know PROC REPORT generally."
The clinical knowledge that makes it "clinical"
This is where the exam separates "SAS programmer" from "clinical trials programmer."
You're expected to understand:
- CDISC SDTM SAS programming concepts and domain models, at least at a working level.
- ADaM dataset creation in SAS, including analysis-ready structures, derivation conventions, timing variables, and flags.
- Clinical trial terminology: adverse events, concomitant medications, endpoints, baseline, treatment-emergent concepts.
- Regulatory submission expectations and validation mindset. Not necessarily deep regulatory law, but the idea that outputs must be reproducible, traceable, and QC'd.
- Medical coding systems like MedDRA and WHO Drug. You don't need to memorize dictionaries, but you need to recognize how coded terms fit into datasets and outputs.
- Protocol-specific derivations. That's the real world. Specs win over "generic best practice."
Where people commonly lose points
A few repeat offenders show up every cycle.
Complex TLF creation with precise formatting's the big one, because candidates answer with "valid SAS" but not "correct deliverable." Another is SAS clinical data validation and QC procedures. People know "double program" exists, but they don't know what to check, what mismatches matter, or how to structure QC logic.
ADaM timing variables and flags also cause trouble. Missing data and partial dates too. Clinical data loves partials. You can't treat "2025-03" like a normal date without thinking through imputation rules, display conventions, and derivation expectations.
Metadata questions can sneak in here as well. Not always heavy, but enough that you should know how metadata informs mapping, derivations, and consistency across domains.
Time pressure and pacing (how to not melt down)
You're balancing speed with accuracy across 60 to 70 questions, and some are long scenarios. Code review items demand attention to detail, and anxiety makes you skim. That's when you miss the one word that changes the whole requirement.
My pacing advice's boring but works:
- First pass: take the quick wins, mark the time traps.
- Second pass: handle the medium scenarios.
- Last pass: wrestle the monsters, but don't donate 8 minutes to one question unless you're sure it's worth it.
Read the question twice. Then read the last line again.
What makes the accelerated version harder
Compressed coverage's the headline, but the real issue's cognitive load. You're asked to hold SAS mechanics, SDTM versus ADaM intent, derivation logic, and output conventions in your head at the same time, and the accelerated format gives you less time to reinforce concepts before moving on.
It assumes you already have baseline SAS competency. It also expects independent learning and rapid application. If you need a slow ramp on macros or PROC SQL, you'll feel behind immediately.
How to make the exam feel easier (practical prep)
Hands-on practice's the difference maker. You want real-ish clinical datasets, not toy "class" tables that never have missing values or messy dates.
A prep stack that works:
- Systematically study CDISC docs (start with SDTM basics, then ADaM concepts).
- Build a personal library of TLF templates you can explain, not just copy.
- Do timed practice tests. Real timer. No pauses.
- Review real-world clinical study reports and mock specs so you get used to the voice and structure.
If you want extra reps, an A00-281 practice test style pack can help you identify weak spots quickly. I mean, I'm not saying it replaces the SAS Institute A00-281 study guide, but it's useful when you need volume and you need it now. I've seen people use an A00-281 Practice Exam Questions Pack to drill pacing, then go back to docs and labs to fix the root issues.
How it compares to other SAS certs
Compared to SAS Base Programming, A00-281's harder because it's specialized and expects clinical judgment, not just syntax and procedures.
It's similar difficulty to SAS Advanced Programming, but the pain's different. Advanced is more "SAS language depth." A00-281 is "SAS plus clinical rules plus deliverable expectations." It's less statistically intense than something like Statistical Business Analyst, but more applied and detail-heavy in a way that feels like real work.
Quick FAQs people ask
How much does the exam cost? Usually a few hundred USD, varies by region and promos, check SAS for current pricing.
What's the passing score? SAS may use scaled scoring and doesn't always make the cut score simple to predict, so focus on readiness, not gaming the number.
Are there official practice tests? Sometimes yes, sometimes limited. If you use third-party options, pick ones that explain answers and reflect current objectives. And if you want a fast drill resource, the A00-281 Practice Exam Questions Pack is one option at $36.99, just pair it with real CDISC reading and hands-on coding so you're not memorizing patterns.
Bottom line: the SAS A00-281 exam's difficult if you're missing either side of the skill set. If you've got solid SAS chops and you've lived in SDTM/ADaM land for a bit, it's challenging but very manageable.
A00-281 Exam Objectives and Content Blueprint
What you're actually signing up for with this exam
The SAS A00-281 exam isn't your typical programming certification. This is the accelerated version of clinical trials programming, which means SAS Institute basically crammed what would normally be two separate exams into one beast of a test. You're expected to know both base SAS programming AND clinical trials-specific stuff, all in one sitting.
Look, if you've been working as a clinical programmer for a year or two, this exam validates what you already do daily. But coming from general SAS programming? You'll need to learn a whole new vocabulary around drug development, regulatory submissions, and those CDISC standards everyone in pharma obsesses over.
The certification proves you can create analysis datasets, generate tables for regulatory submissions, and follow the quality control practices that keep drugs from getting rejected by the FDA. It's a lot more specialized than the general SAS Base Programming exam.
Breaking down the official blueprint percentages
SAS Institute publishes domain weightings for this exam, and they shift around a bit with each update. Clinical trials programming foundations usually account for 15-20% of questions. That's your basic understanding of study phases, what a CRF actually is, and how clinical programmers fit into the drug development timeline.
Clinical data standards and structures take up the biggest chunk at somewhere around 20-25% of the exam. This is where CDISC becomes your best friend or worst enemy. You need to know SDTM domains inside and out. DM, AE, CM, VS, LB, EX.. these aren't just random letter combinations. They're the building blocks of every clinical trial dataset you'll ever create.
Data manipulation and analysis dataset creation is the meatiest section at 25-30%. This is where your actual SAS programming chops get tested. Can you merge domains correctly? Can you calculate study days without messing up when subjects have gaps in treatment? Can you flag treatment-emergent adverse events using the right logic?
Short answer: it's tough.
Tables, listings, and figures programming hits around 20-25%. PROC REPORT becomes your go-to tool here, though PROC SQL gets plenty of love too. You'll need to create demographic tables, AE summaries by system organ class, lab shift tables. All the standard TLFs that show up in every clinical study report.
Validation, quality control, and documentation rounds out the exam at 10-15%. Smaller percentage, but critical stuff. How do you validate your programs? What documentation do you need? How do you trace derivations back to source data?
The clinical trials foundation knowledge nobody tells you about
Before you write a single line of SAS code, you need context. What's the difference between Phase I and Phase III? Why does it matter? Phase I trials are small, usually healthy volunteers, testing basic safety. Phase III trials are huge. Thousands of patients, proving the drug actually works better than existing treatments.
Understanding CRFs matters more than you'd think. These paper or electronic forms capture every data point from a clinical trial. Blood pressure readings, adverse events, medication dosages. Everything flows from CRFs into your SAS datasets. If you don't understand how data gets collected, you'll make wrong assumptions when programming.
The regulatory environment shapes everything we do. FDA and EMA have specific requirements for data submissions. Define.xml files, analysis traceability, validation documentation. These aren't suggestions. They're requirements that can delay or kill a drug approval if you mess them up.
Good Clinical Practice principles apply to programming too, not just clinical sites. You need to understand concepts like informed consent, adverse event reporting timelines, and protocol deviations because they all affect your data and programming logic.
I once spent three days debugging a program only to realize the issue wasn't my code at all but a protocol amendment that changed how we defined the analysis population. Nobody had updated the specs. That kind of thing happens more often than it should in this field.
CDISC standards are non-negotiable in this field
SDTM (Study Data Tabulation Model) is the format FDA expects for raw clinical data. Each domain has a specific structure. DM contains demographics, one record per subject. AE contains adverse events with multiple records per subject, one per event. VS captures key signs, LB handles laboratory results, EX tracks drug exposure.
The implementation guide isn't light reading, but you need to know it. Variable naming conventions, controlled terminology, required versus expected versus permissible variables. When is AESTDTC required versus AESTDT? Why do some domains need --SEQ variables while others don't?
Parent-child relationships between domains matter for programming. SUPPQUAL domains hold additional qualifiers. RELREC datasets define relationships that don't fit standard structures. If you're creating SDTM from scratch, you'll deal with these constantly.
ADaM (Analysis Data Model) sits on top of SDTM. ADSL is your subject-level dataset, one record per subject with all the demographic and treatment variables you need for analysis. BDS handles findings data like lab results and key signs in a one-record-per-timepoint structure. OCCDS manages occurrence data like adverse events.
Traceability requirements mean every derived variable in ADaM needs clear documentation back to SDTM. How did you calculate AVAL? Where did BASETYPE come from? This isn't optional. It's part of the regulatory submission package.
Data manipulation techniques that actually show up on the exam
Reading raw clinical data sources involves more than a simple DATA step. You're dealing with multiple file formats, external databases, inconsistent date formats, and missing data patterns that would make a general programmer cry.
Partial dates are everywhere in clinical trials. A patient remembers starting a medication in March 2023 but can't recall the exact day. How do you handle that? Convention says you impute conservatively. Latest possible start date when calculating duration, earliest possible date when determining if an event was treatment-emergent.
Study day calculations trip up tons of programmers. Study day 1 is the first day of treatment. There's no study day zero. Days before treatment are negative. But what if treatment starts and stops multiple times? What if there's a gap? The logic gets complex fast.
Baseline identification isn't always "last measurement before treatment." Sometimes it's "last non-missing measurement within 30 days before first dose." Sometimes it's "average of all pre-treatment measurements." Protocol-specific rules matter, and you need to implement them correctly.
Treatment-emergent adverse event flagging follows specific rules. Generally, an AE is treatment-emergent if it starts on or after first dose and within some number of days after last dose. But what about events that start before treatment but worsen during treatment? That's treatment-emergent too, just with different logic. Kind of a gray area sometimes.
TLF programming separates junior from senior programmers
PROC REPORT is the workhorse for clinical tables. DEFINE statements set up your columns, COMPUTE blocks calculate statistics or format values, spanning headers organize related columns. You need to know the difference between DISPLAY, ORDER, ANALYSIS, and COMPUTED variables.
It's actually pretty intuitive once you practice.
Demographic tables seem simple until you need to handle multiple treatment groups, add statistical tests, format percentages consistently, and deal with missing data appropriately. Should you show N=0 categories? How do you align decimal points across different statistics?
Adverse event tables by system organ class and preferred term require sorting by frequency, most common events first. Calculate percentages correctly where denominator is subjects in treatment group, not total AEs. Handle the MedDRA hierarchy. You can't just sort alphabetically and call it done.
Lab shift tables show how many subjects moved from normal at baseline to low, normal, or high post-baseline. Creating these requires merging baseline values with post-baseline values, categorizing based on reference ranges, and presenting results in a readable format. The advanced programming skills from A00-212 definitely help here.
Validation and QC practices that prevent disasters
Double programming is standard in clinical trials. An independent programmer recreates key outputs, and you compare results. Discrepancies get investigated. This catches logic errors, typos, and misunderstandings before they reach regulators.
Documentation requirements go beyond comments in your code. You need specification documents describing what each program does. Validation plans explaining how outputs will be checked. Validation reports documenting that checks were completed successfully.
QC checklists help ensure consistency. Did you check for duplicate records? Verify date formats? Confirm treatment group assignments match randomization? These mundane checks catch embarrassing errors. Trust me, I've seen it happen more than once in my career.
Why the blueprint changes and what that means for you
SAS Institute updates exam objectives periodically as the industry evolves. CDISC releases new implementation guide versions. FDA updates expectations for electronic submissions. The exam adapts to reflect current practices, not what was standard five years ago.
Before you start studying, download the current blueprint from SAS Institute's website. Don't rely on someone's blog post from 2019 or study materials from 2021. The weightings shift, topic emphasis changes, and new content gets added.
I've seen people prepare using outdated materials and get blindsided by questions on topics that weren't previously emphasized. Check the publish date on everything you study. If it predates the current exam version, use it cautiously.
How this exam connects to the broader clinical programming path
The A00-281 accelerated version combines content from the standard A00-280 clinical trials exam with additional advanced topics. It's designed for experienced SAS programmers who want to pivot into clinical trials without taking multiple sequential exams.
If you're already strong in SAS programming fundamentals, the clinical-specific content becomes your focus. But if you're shaky on DATA step processing, macro programming, or PROC SQL, you'll struggle with the accelerated pace.
The certification opens doors in pharmaceutical companies, CROs (contract research organizations), and regulatory agencies. Clinical programmers are in demand, and this credential proves you can handle the specialized requirements of drug development work.
Conclusion
Wrap up
Look, the SAS A00-281 exam isn't something you just walk into cold. Real deal here.
It's really testing whether you can execute clinical trials programming work, not memorizing syntax like some parrot but actually applying CDISC SDTM SAS programming principles, constructing ADaM dataset creation in SAS workflows, and delivering clinical listings tables figures (TLFs) SAS that regulatory reviewers won't bounce back with red flags all over them. I mean, if you've spent time in the pharma space or clinical data world, you already know this stuff matters. A00-281 certification signals that you understand the accelerated track demands, which honestly separates weekend warriors from people who can handle production environments.
Your study plan matters more than how long you've used SAS. Here's the thing. Some folks with five years of general SAS experience absolutely bomb this exam because they never touched clinical trial data programming or CDISC standards. Others with 18 months of focused clinical work pass because they've done actual SAS clinical data validation and QC in the real world. The exam tests proc report proc sql clinical programming patterns you'll use daily. Derivations, metadata handling, validation loops. Spend time on hands-on work, not just reading slides.
Practice tests? That's where you figure out what you don't know.
Official resources help, but third-party materials give you volume and variety you need to spot knowledge gaps. When you're reviewing missed questions, treat it like a QC check. Dig into why your logic failed, trace it back to the exam objectives, then rebuild your mental model. That's how you turn weak areas into strengths before exam day. My old manager used to say the best programmers aren't the ones who never make mistakes, they're the ones who catch their own errors before anyone else sees them. Same principle applies here.
The SAS Clinical Trials Programming Using SAS 9 certification isn't just resume decoration. It's proof you can handle the technical and regulatory demands of clinical programming. Honestly, whether you're trying to break into the field or validate skills you've been using for years, passing the SAS A00-281 exam opens doors at CROs, pharma companies, and biotech startups that need people who actually know this domain. People who can apply it under pressure.
If you're serious about passing the SAS clinical trials programming exam on your first attempt, you need realistic practice under exam conditions. The A00-281 Practice Exam Questions Pack at /sas-institute-dumps/a00-281/ gives you that. Questions that mirror the actual exam format, detailed explanations that teach you why answers work, and enough repetitions to build confidence. Not gonna lie, that's the difference between walking in prepared versus hoping you studied the right stuff.
Show less info
Comments
Hot Exams
Related Exams
Accredited Financial Examiner
IBM AIX v7 Administrator Specialty
Marketing Cloud Advanced Cross Channel Accredited Professional Exam
SAP Certified Technology AssociateSAP HANA Cloud Provisioning and Administration
PSE Palo Alto Networks System Engineer Professional - Prisma Cloud
SAS Clinical Trials Programming Using SAS 9 - Accelerated Version 9 Accelerated Version
Administering SAS Viya 3.5
SAS Base Programming for SAS 9
SAS Certified Associate: Programming Fundamentals Using SAS 9.4
SAS Viya 3.5 Natural Language Processing and Computer Vision
SAS Advanced Programming for SAS 9
A00-250 SAS Platform Administration for SAS9
SAS BI Content Developmentfor SAS 9
SAS Data Integration Development
Clinical Trials Programming Using SAS 9
SAS Statistical Business Analysis SAS9: Regression and Model
How to Open Test Engine .dumpsarena Files
Use FREE DumpsArena Test Engine player to open .dumpsarena files

DumpsArena.co has a remarkable success record. We're confident of our products and provide a no hassle refund policy.

Your purchase with DumpsArena.co is safe and fast.
The DumpsArena.co website is protected by 256-bit SSL from Cloudflare, the leader in online security.